Process Validation Including Software | MDV-Solve
Process Validation
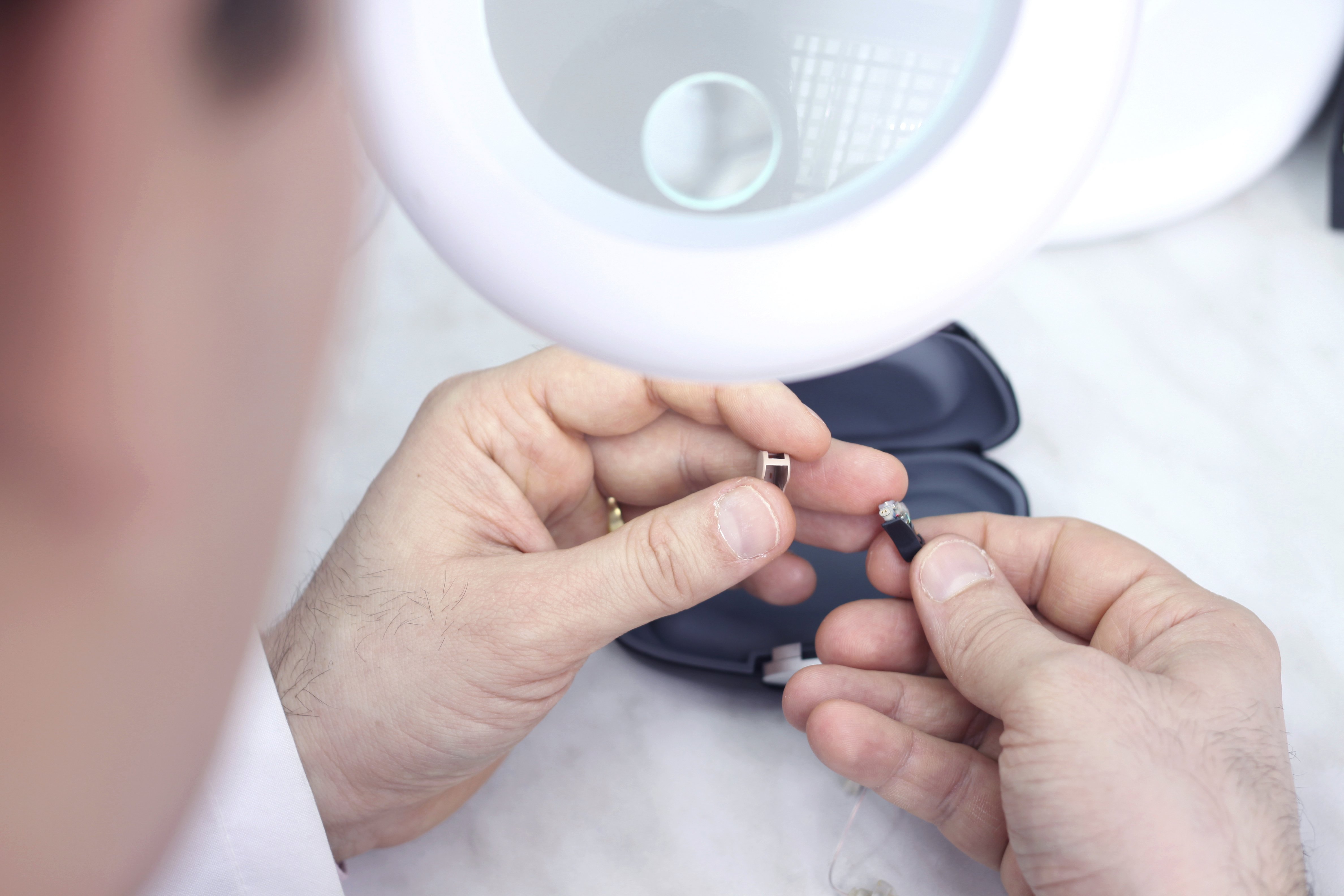
Process validation establishes scientific evidence that a process is capable of consistently delivering quality product. We guide you through IQ, OQ, and PQ.
Services offered:
- Master Validation Plan
- Installation Qualification (IQ)
- Operational Qualification (OQ)
- Performance Qualification (PQ)
- Software validation (per IEC 62304 for medical device software)
Related Services
The New EU IVD Regulation (IVDR)
The In Vitro Diagnostic Regulation stipulates all requirements IVD manufacturers must comply with.
+ Learn MorePost-Market Surveillance and Vigilance
Establishment and implementation of PMS to review experience gained from devices in the post-production phase.
+ Learn MoreQuality System under EN ISO 13485, US FDA QSR - GMP
The purpose is to enable you to establish and implement an effective tailored QMS system …
+ Learn More